By mapping transmission and identifying groups of people at risk of HIV infection, a study examining HIV cases reported in Victoria between 2000 and 2020 illustrates a new approach that could make public health efforts more timely and effective.
In a study that Lancet Western Pacific Regional Health, Researchers used a technique called molecular epidemiology to better understand the transmission of HIV-1, the most common type of human immunodeficiency virus (HIV), in Victoria. By linking the genetic data of the virus with traditional epidemiological information, the researchers identified transmission clusters among people infected with HIV-1. These findings can help improve public health strategies to reduce HIV-1 transmission rates.
Dr George Taiaroa of the University of Melbourne, a research fellow at the Doherty Institute and first author of the study, said that just as cartographers create detailed maps to navigate landscapes, researchers can create genetic maps to better understand how viruses move through populations, and map pathways. Transmission and prevalence
“Strategies using these approaches can be valuable in guiding public health responses, thereby providing an evidence base and foundation for those considering adopting these methods routinely in Australia,” Dr Taiaroa said.
Several smaller transmission groups indicated possible transmitted drug resistance (TDR) to antiretroviral therapies. These treatments include treatments that prevent the virus from maturing and becoming infectious, as well as treatments that prevent the virus from replicating.
Dr. Taiaroa added, “Identifying transmitted drug resistance can allow for more targeted selection of antiviral drugs.”
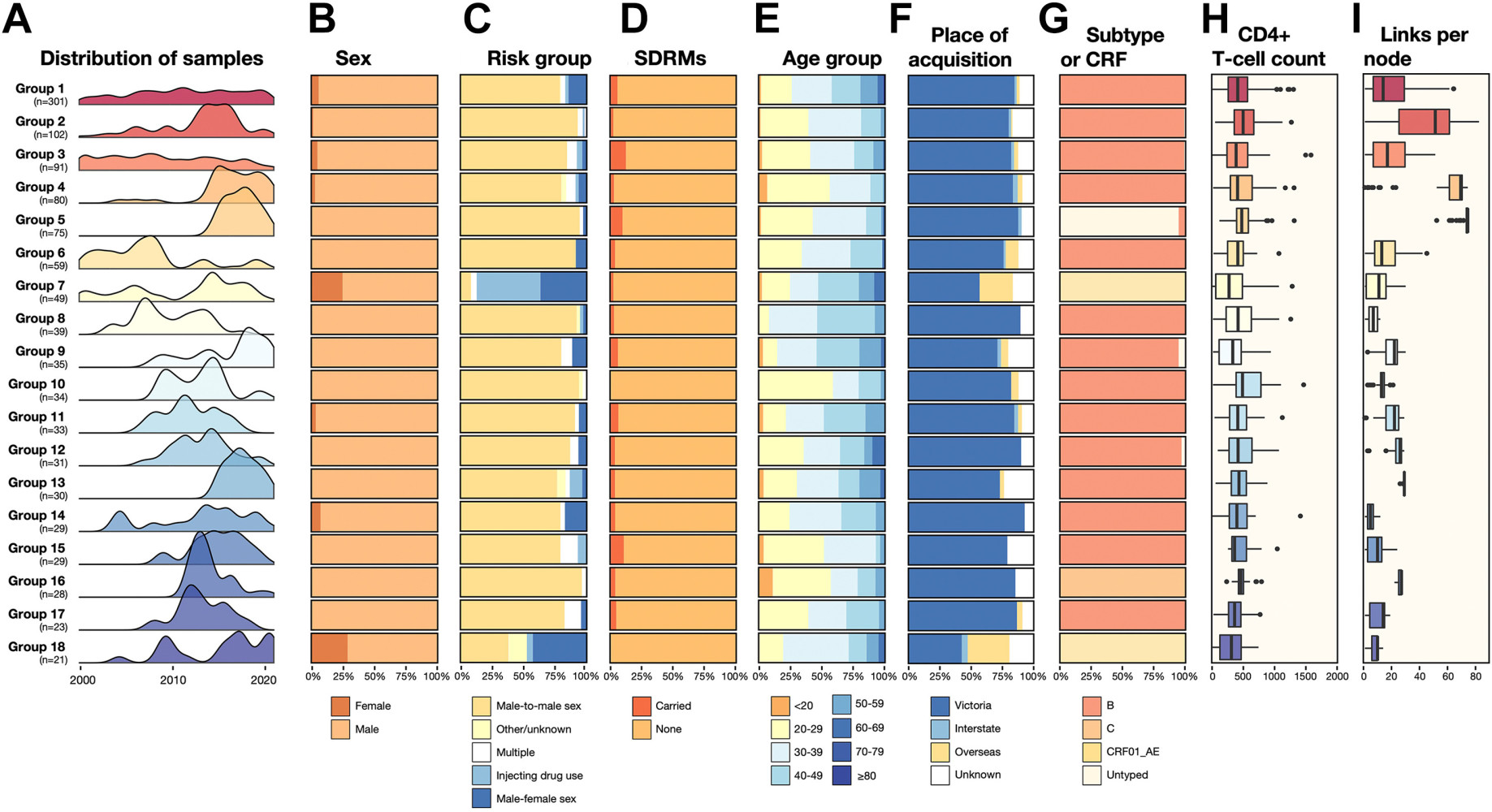
Characteristics of large clusters of HIV-1 transmission in Victoria, Australia. (a) Distribution of samples for molecular transport groups. B–E) Summary of the proportions of different genders (B), risk groups (C), SDRM (D), age groups (E), possible site of acquisition (F) and subtype or CRF (G) in the main groups the transfer . (H) Distribution of the number of CD4+ T cells in the main transfer groups. Boxplots show the median and IQR, with whiskers representing the highest and lowest values within 1.5 × IQR of the upper and lower quartiles, and dots representing outliers. (I) Distribution of links of each node in the main transmission groups. Box plots show median and interquartile range (IQR), formatted as above.
Australia has a relatively low rate of new HIV infection compared to many high-income countries, thanks to leading global care models for the prevention and management of HIV infection. However, disruptions in healthcare caused by the COVID-19 pandemic have set back some progress, and higher rates of infection continue to disproportionately affect Aboriginal and Torres Strait Islander people and migrant communities.
Professor Sharon Levin of the University of Melbourne, director of the Doherty Institute, an international HIV expert and lead author of the paper, said that as Australia aims to further reduce HIV transmission, the integration of molecular approaches may play an important role while respecting the complex ethical framework. . To manage genetic data
We work closely with affected communities to understand the evidence on the acceptability of these approaches in Australia. Ensuring informed consent and addressing potential stigma are essential steps, Professor Levin said.
Importantly, this work involved significant engagement with community groups and stakeholders around the world, including the National Association of People Living With HIV Australia (NAPWHA), Living Positive Victoria and the Melbourne Center for Sexual Health.
Richard Kane, CEO of Living Positive Victoria, praised the Doherty Institute obligation To interact with the community
The efforts undertaken by the Doherty Institute over the past two years to prioritize the meaningful participation of people living with HIV in the potential implementation of HIV phylogenetic tracing in Victoria provide a model for a truly participatory approach, one that seeks ethical and potential scrutiny. The legal implications of using this emerging technology, said Mr. Kane.
Doherty researchers regularly attend state and national meetings with community groups to share research methodology and findings, hear participants’ views, address potential concerns, and discuss what is needed to use this approach appropriately and effectively in Australia. to discuss
“As CEO of Living Positive Victoria and a person living with HIV, understanding that the introduction of new technologies may create potential barriers to testing and trusting the institutions implementing it, for the community to understand its role in Greater measures of empowerment are necessary, Mr Kane added, to move towards the virtual elimination of HIV in Australia.
“Molecular epidemiology is an exciting new technology that has great potential to assist Australia’s HIV response to achieve the virtual elimination of HIV transmission by 2030,” said NAPWHA Chief Executive Aaron Cogle.
“Throughout this study, the Doherty Institute has provided an impressive example of how people living with HIV can be consistently and meaningfully engaged at the national and state/regional level. They have facilitated discussion on how to safely perform real-time phylogenetic analyzes within an appropriate ethical framework and in a decriminalized environment.
“NAPWHA looks forward to continuing this approach with Doherty as we support a policy environment that allows us to safely reap the maximum benefits of HIV phylogenetic tracing in Australia,” Mr Kugel added.
The appropriate introduction of molecular epidemiology for HIV in Australia, with the help and support of the community, has great potential to enhance public health efforts with the findings of this study identifying how this could be a transformative approach to public health.
Detailed review: Taiaroa G, et al. Characterizing HIV-1 transmission in Victoria, Australia: a molecular epidemiological study. Lancet Western Pacific Regional Health, (2024). https://doi.org/10.1016/j.lanwpc.2024.101103
Cooperation: University of Melbourne, Department of Health Victoria, Alfred Health, Life Positive Victoria, Melbourne Center for Sexual Health, Monash University, National Association of People Living with HIV Australia (NAPWHA).
Funds: National Health and Medical Research Council, Australian Research Council
#Innovative #genetic #analysis #maps #HIV #transmission #Victoria #meaningful #community #engagement